What Is the True Identity of Microfold Cells?
- 演化之聲

- Mar 11
- 4 min read
Within the epithelial lining of the human small intestine exists a specialized cell type known as the microfold cell (also called the M cell). For many years, the biological identity of these cells has remained somewhat mysterious. They are typically found in the follicle-associated epithelium that overlies Peyer's patches. Traditionally, microfold cells were thought to function merely as transporters, carrying antigens from the intestinal lumen to immune cells located beneath the epithelium. However, a study published near the end of 2025 has provided a deeper understanding of their role.

To investigate the identity of microfold cells, researchers cultured human intestinal stem cells (LGR5⁺ intestinal stem cells). These stem cells developed into human intestinal organoids containing small three-dimensional crypt- and villus-like structures. By adding differentiation factors to the culture medium—specifically the proteins RANKL, TNF, and retinoic acid—some cells within the organoids began differentiating into GP2⁺ microfold cells. Using single-cell RNA sequencing, the researchers were able to clearly identify the various cell types present within the organoids, including several intestinal epithelial cell types, transitional precursor cells leading toward microfold cells, and fully differentiated microfold cells. This relatively simple organoid system allowed the investigators to precisely trace the genetic trajectory of microfold cell differentiation.

During this differentiation process, the expression patterns of three genes—SPIB, ICAM2, and GP2—served as markers for the developmental stages of microfold cells. Based on these markers, the cells could be classified into early (SPIB⁺ ICAM2⁻), intermediate (SPIB⁺ ICAM2⁺ GP2⁻), and mature (SPIB⁺ ICAM2⁺ GP2⁺) stages. Genomic analysis revealed that more than 1,600 genes changed significantly during maturation. As differentiation progressed, characteristics typical of intestinal epithelial cells gradually diminished, while genes associated with immune interactions became increasingly prominent.
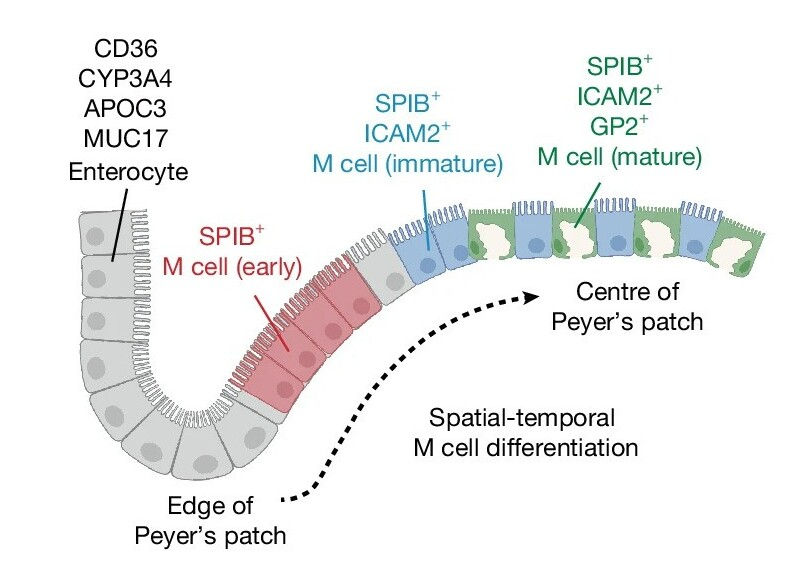
Remarkably, the gene expression profile of mature microfold cells showed extensive overlap with that of dendritic cells, a key type of immune cell. Several classical dendritic-cell marker genes—including CD83, LAMP3, IL7R, and FSCN1—became progressively upregulated during microfold cell maturation. In addition, the transcription factors SPIB and RUNX2, both known to participate in dendritic-cell development, were shown to be essential regulatory factors for human microfold cells. Together, these findings point to a new concept: although microfold cells originate from intestinal stem cells rather than hematopoietic stem cells, their regulatory gene networks converge toward those typical of immune cells, and their behavior resembles that of dendritic cells.
A key piece of evidence supporting this idea is that microfold cells express MHC-II molecules, a protein complex normally associated with antigen presentation by immune cells. Even in the absence of inflammatory stimulation, human microfold cells naturally express MHC-II along with the antigen-processing factor CD74, which appears on the basolateral cell membrane. In contrast, ordinary intestinal epithelial cells only begin expressing MHC-II after stimulation by interferon-γ, and even then the levels are far lower than those observed in immune cells or microfold cells. Electron microscopy further revealed multiple forms of intracellular MHC-II transport structures within microfold cells, including multivesicular and lamellar compartments, closely resembling those found in genuine dendritic cells. These observations indicate that microfold cells possess a complete system for antigen processing and transport.
If microfold cells express MHC-II, they should also be capable of presenting antigens. To test this, researchers co-cultured organoids containing microfold cells with T cells (using the Jurkat cell line). They then introduced the immunogenic 33-mer peptide derived from α2-gliadin, a peptide known to interact with the MHC-II molecule HLA-DQ2.5. The experiments demonstrated that only microfold cells capable of expressing HLA-DQ2.5 could efficiently load and present this peptide to T cells, triggering T-cell activation. These results show that microfold cells can function as antigen-presenting cells rather than merely acting as a bridge for antigen transport. This antigen-presenting ability may also help explain why gluten consumption can trigger coeliac disease.

The study also revealed a striking difference between humans and mice. In mice, microfold cells do not express MHC-II and lack the capacity for antigen presentation. This species difference may explain why earlier mouse models failed to detect the antigen-presentation role of microfold cells.
Overall, this research clarifies the fundamental nature of microfold cells and opens new perspectives for understanding intestinal immunity and developing therapeutic strategies for related diseases.
Author: You-Ye Shui
Reference:
1. Wang D et al. (2025). Human gut M cells resemble dendritic cells and present gluten antigen. Nature.
2. Kim SH and Jang YS .(2017). The development of mucosal vaccines for both mucosal and systemic immune induction and the roles played by adjuvants. Clin Exp Vaccine Res.
3. Velazquez-Soto H et al. (2022). Historical evolution, overview, and therapeutic manipulation of co-stimulatory molecules. World J Immunol.




Comments