A Silurian Leech Fossil Reshapes the Evolutionary History of Leeches
- Rodrigo

- Mar 11
- 5 min read
Updated: Apr 11
Leeches are soft-bodied worms with slick, flexible bodies, well known for their blood-feeding habits. They commonly inhabit wetlands and damp forest floors. Long before humans began recording history, our ancestors had already encountered these persistent annelids.
For hundreds of millions of years, leeches have appeared almost unchanged in both form and lifestyle. Their familiar appearance—a soft-bodied worm equipped with suckers and capable of parasitism—gives the impression of a biological "living fossil," as though they had existed in essentially their modern form since the beginning. But is this assumption truly correct?
In 2025, a remarkable fossil discovered in the Waukesha Lagerstätte of Wisconsin, USA, offered an important clue to this long-standing question. The fossil comes from the Telychian stage of the Silurian Period, approximately 437 million years ago, and represents a leech-like animal named Macromyzon siluricus. The genus name derives from the Greek words macro ("large") and myzon ("sucker"), referring to the prominent sucker at the rear of the body, while the species name refers to the Silurian age of the fossil.
The body of Macromyzon siluricus measures about 51 millimeters in length. Its outline is worm-like to sublanceolate in shape, with a truncated anterior end. The body gradually widens toward the rear, reaching its maximum width slightly before the posterior margin. The width of the posterior end is roughly twice that of the anterior end. The body is clearly segmented, and each segment is subdivided into six ring-like annuli arranged in regular patterns. At the very end of the body lies a large caudal sucker.
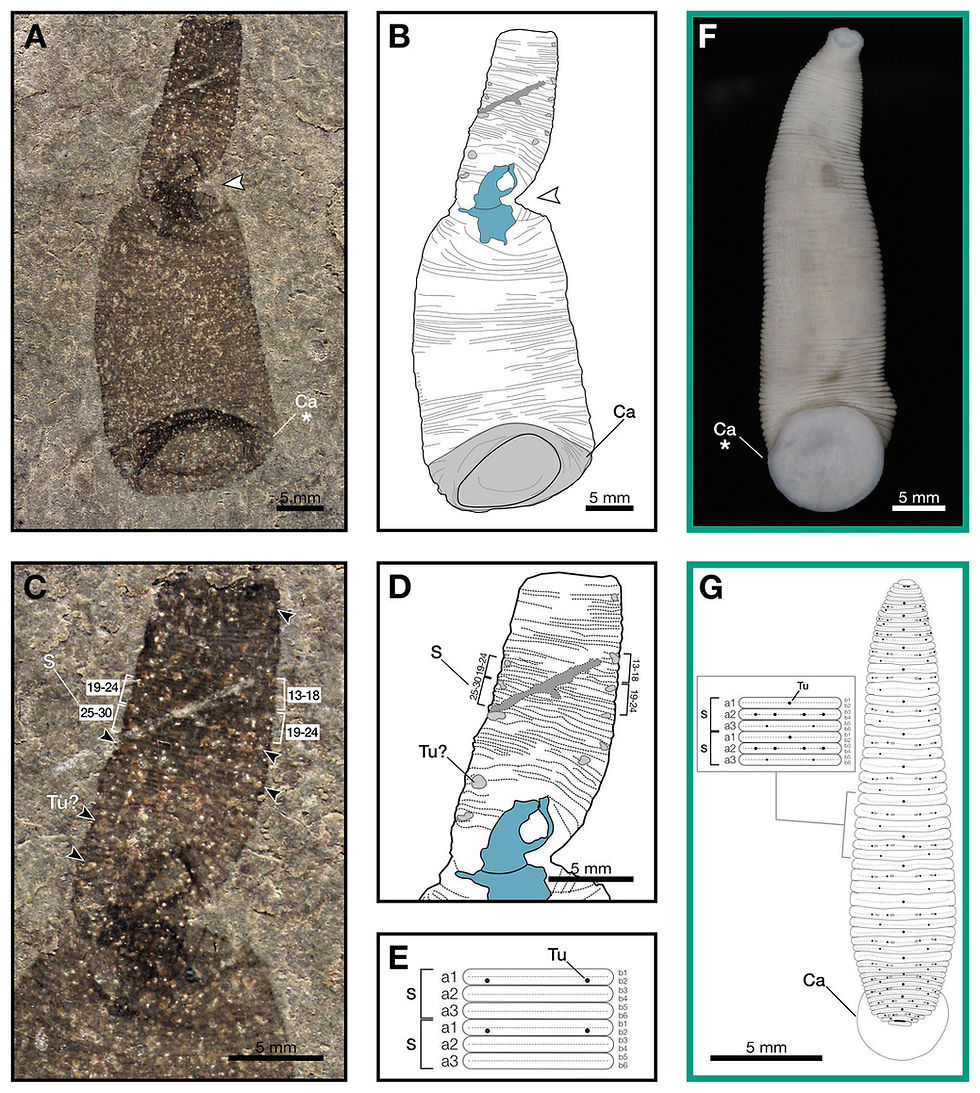
Unlike previously known leech fossils, this specimen preserves exceptionally detailed soft tissues. Leeches consist almost entirely of soft tissue, which decays rapidly after death. Because of this, paleontologists have historically struggled to find body fossils of leeches in the geological record. Instead, researchers have had to rely on indirect forms of evidence to infer their evolutionary history.
One method uses molecular genetics. By comparing DNA differences among living leeches and other annelids, scientists estimate divergence times through molecular clock models. Another line of evidence comes from fossil cocoons—gelatinous egg cases secreted by clitellate annelids during reproduction to protect fertilized eggs.

Both sources of evidence have clear limitations. Molecular clock estimates depend heavily on assumptions about mutation rates and calibration points, which can strongly influence the resulting timelines. Cocoon fossils can confirm that some clitellate organism once existed, yet they cannot reveal whether the animal was an earthworm, an early leech, or an intermediate form that had not yet fully diverged. They also provide no direct information about the animal's anatomy or ecology.

This is precisely why the Waukesha fossil is so significant. It preserves a combination of features that unmistakably resemble modern leeches: an elongated body, finely subdivided segments, and a prominent posterior sucker. At the same time, it retains several primitive characteristics. These features indicate that the animal was not a modern leech itself, but rather a stem-group leech situated near the base of the leech evolutionary tree.
The implications of this discovery extend far beyond identifying a single ancient species. The fossil fundamentally reshapes the evolutionary timeline of leeches. Molecular clock estimates previously suggested that true leeches originated between roughly 230 and 140 million years ago, most likely during the Jurassic or Cretaceous. Yet Macromyzon siluricus, preserved in Silurian deposits dated to about 437 million years ago, predates these estimates by more than 200 million years.
This indicates that the fundamental leech body plan—including the development of suckers and specialized segmentation—had already emerged in marine environments during the Paleozoic era, far earlier than previously thought.

In other words, the blood-sucking leeches we encounter today in wetlands and forests are not creatures that appeared suddenly in evolutionary history. They are the product of a long evolutionary journey. Their ancestors likely originated in ancient seas, gradually colonized freshwater environments and eventually terrestrial habitats, and only later evolved the specialized blood-feeding lifestyles that make them so familiar today.
Author: Rodrigo
Reference:
de Carle, D., Iwama, R. E., Wendruff, A. J., Babcock, L. E., & Nanglu, K. (2025). The first leech body fossil predates estimated hirudinidan origins by 200 million years. PeerJ, 13, e19962. https://doi.org/10.7717/peerj.19962




Comments